
The apparatus is assembled as in the diagram. For this reason, ethanol cannot be completely purified by direct fractional distillation of ethanol-water mixtures. In this example, a mixture of 96% ethanol and 4% water boils at 78.2 ☌ (172.8 ☏) the mixture is more volatile than pure ethanol. Some mixtures form azeotropes, where the mixture boils at a lower temperature than either component. So, by heating the mixture, the most volatile component (ethanol) will concentrate to a greater degree in the vapor leaving the liquid. It is used to refine crude oil.įractional distillation in a laboratory makes use of common laboratory glassware and apparatuses, typically including a Bunsen burner, a round-bottomed flask and a condenser, as well as the single-purpose fractionating column.Īs an example, consider the distillation of a mixture of water and ethanol. If the difference in boiling points is greater than 25 ☌, a simple distillation is typically used. Generally the component parts have boiling points that differ by less than 25 ☌ (45 ☏) from each other under a pressure of one atmosphere. Chemical compounds are separated by heating them to a temperature at which one or more fractions of the mixture will vaporize. JSTOR ( November 2022) ( Learn how and when to remove this template message)įractional distillation is the separation of a mixture into its component parts, or fractions.Unsourced material may be challenged and removed.įind sources: "Fractional distillation" – news Please help improve this article by adding citations to reliable sources. Can you explain this answer? tests, examples and also practice Class 12 tests.This article needs additional citations for verification. than nitrogend)oxygen has a lower density than nitrogen.Correct answer is option 'C'. Can you explain this answer? theory, EduRev gives you anĪmple number of questions to practice It is possible to obtain oxygen from air by fractional distillation because a)oxygen is in a different group of the periodic table from nitrogenb)oxygen is more reactive than nitrogenc)oxygen has higher b.p. Can you explain this answer? has been provided alongside types of It is possible to obtain oxygen from air by fractional distillation because a)oxygen is in a different group of the periodic table from nitrogenb)oxygen is more reactive than nitrogenc)oxygen has higher b.p. Can you explain this answer?, a detailed solution for It is possible to obtain oxygen from air by fractional distillation because a)oxygen is in a different group of the periodic table from nitrogenb)oxygen is more reactive than nitrogenc)oxygen has higher b.p. 
It is possible to obtain oxygen from air by fractional distillation because a)oxygen is in a different group of the periodic table from nitrogenb)oxygen is more reactive than nitrogenc)oxygen has higher b.p. Can you explain this answer? defined & explained in the simplest way possible. Here you can find the meaning of It is possible to obtain oxygen from air by fractional distillation because a)oxygen is in a different group of the periodic table from nitrogenb)oxygen is more reactive than nitrogenc)oxygen has higher b.p. Can you explain this answer? covers all topics & solutions for ClExam.įind important definitions, questions, meanings, examples, exercises and tests below for It is possible to obtain oxygen from air by fractional distillation because a)oxygen is in a different group of the periodic table from nitrogenb)oxygen is more reactive than nitrogenc)oxygen has higher b.p. Information about It is possible to obtain oxygen from air by fractional distillation because a)oxygen is in a different group of the periodic table from nitrogenb)oxygen is more reactive than nitrogenc)oxygen has higher b.p. The Question and answers have been prepared Can you explain this answer? for Clis part of Class 12 preparation. 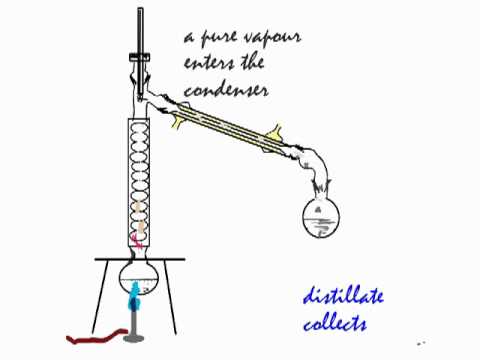
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
